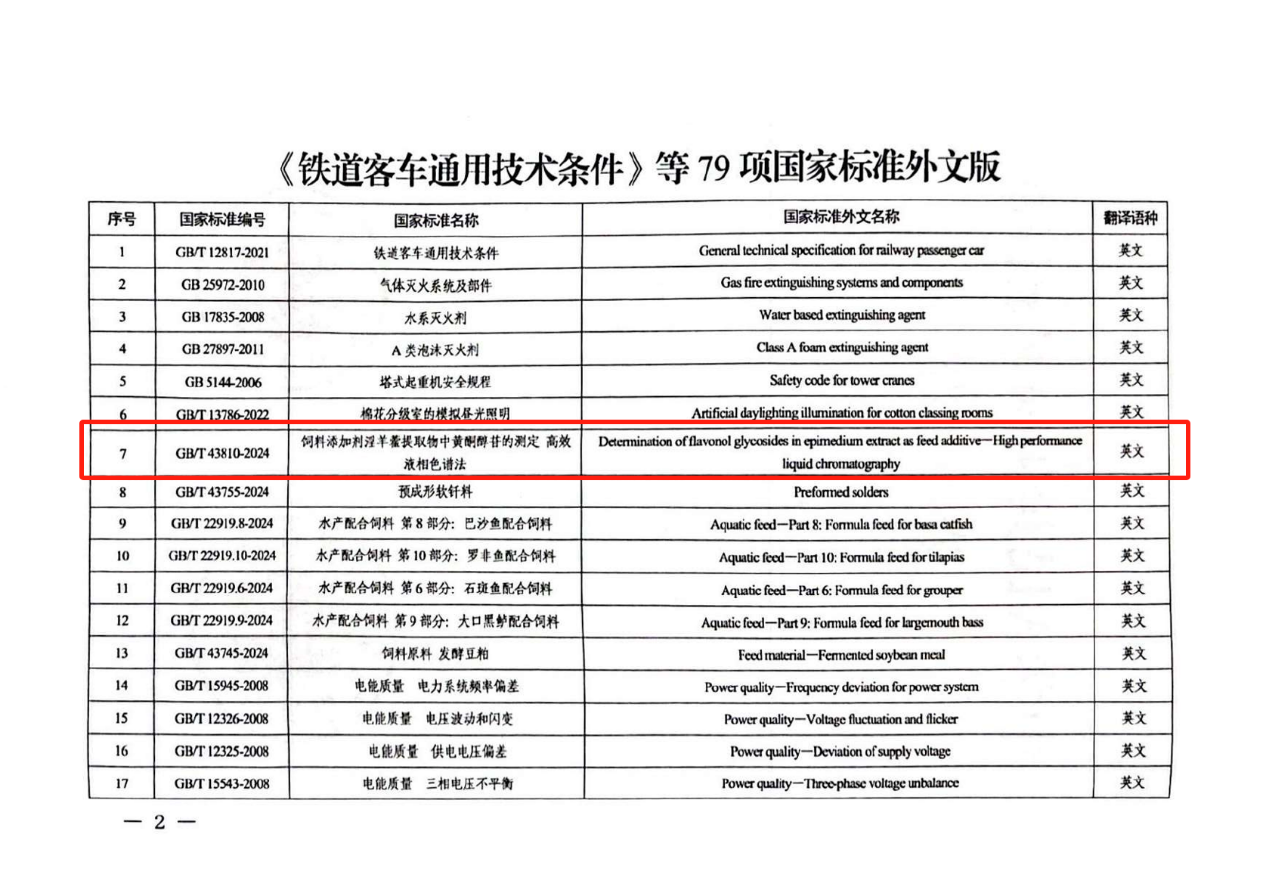
Recently, the English version of the national standard “Determination of flavonol glycosides in epimedium extract as feed additive — High performance liquid chromatography” (GB/T 43810-2024), was officially approved by the State Administration for Market Regulation. Developed and translated by the Functional Livestock Product Team of Institute of Animal Sciences, Chinese Academy of Agricultural Sciences. This standard establishes a recommended benchmark for the detection and quality control of Epimedium extract. The Chinese version of this standard has been promulgated and implemented since October 1, 2024.
Epimedium extract, an important "antibiotic alternative" product and a feed additive listed in the “Feed Additives Catalogue”, has faced market challenges due to significant variations in the composition and concentration of its active ingredients. These inconsistencies stem from species diversity, differing production processes, and the lack of standardized detection methods. To standardize regulation and ensure safe application in the livestock industry, the research team aimed to address the issues of inconsistent detection methods and the lack of representativeness in the quality control components of Epimedium extract. They spearheaded the development of this national standard, which details a high-performance liquid chromatography (HPLC) method for determining five flavonol glycosides in Epimedium extract feed additives: icariin, sagittatoside A, sagittatoside B, sagittatoside C, and baohuoside I.
The implementation of this standard are significant for improving the quality standard system of plant extract feed additives and promoting high-quality development in China’s livestock, pharmaceutical, and related industries. Moreover, the approval of the international version serves as a bridge to facilitate the standardized use of Epimedium extract feed additives, allowing international stakeholders to align with China’s quality standards and providing technical support to related global industries.